|
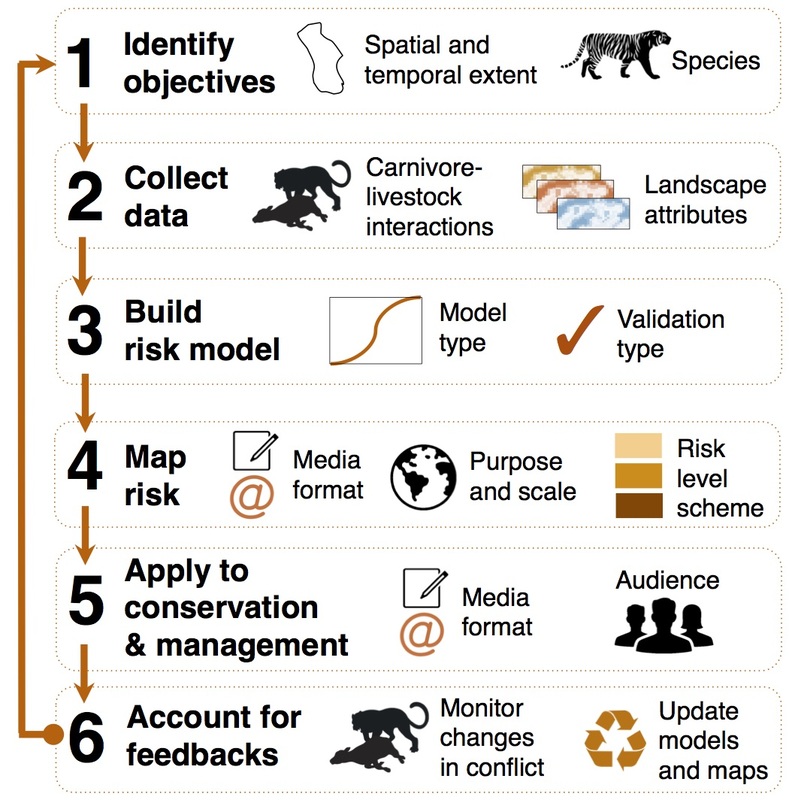
-- Jennie Miller, PhD ‘15 One of the biggest challenges for a wild carnivore today is avoiding conflict with humans. With livestock now using 30% of ice-free land on our planet, most carnivores likely live within or adjacent to a buffet of domestic animals, served up fear-free, tender and ready for the taking. But this livestock buffet has hidden costs when owners use lethal control or poison carcasses to kill carnivores and reduce future livestock losses. Livestock owners and wildlife managers have struggled for centuries to protect domestic animals and carnivores alike by implementing a variety of nonlethal tools to reduce attacks, but knowing when and where to focus tools is tricky. Last week Biodiversity and Conservation published my review on an emerging method for mapping livestock depredation hotspots, which aims to strengthen efforts to reduce human-carnivore conflict. The article describes spatial risk models how they have been used by conservationists in outreach with livestock owners, managers and policymakers. For instance, in rural Tanzania, the Ruaha Carnivore Project shared risk maps with livestock owners through PowerPoint presentations conveyed in Swahili and delivered during educational movie nights to help people recognize distributions of predator risk in their area. In Gujarat, India, risk maps on Asiatic lion attacks on livestock were included in conflict management manuals distributed to high-risk villages. In Hunchun, China, hotspot maps were included in a policy document that reportedly contributed to the government agency increasing efforts to resolve human–tiger conflict. In other words, maps are an ideal tool for communicating information with non-scientist audiences. And with increasing access to the internet and smartphone technology across the world, risk maps can be shared with stakeholders in innovative ways. A great example is the interactive online map of wolf risk to livestock in Wisconsin, created by the Carnivore Coexistence Lab at the University of Madison-Wisconsin, where ranchers can type in their address and view to risk over several years around their property. Below are a few samples of the variety of visual forms that risk maps can take. Especially if information already exists on the when and where of livestock kills, models can be relatively simple and inexpensive to integrate into the conservation decision-making process. In areas where financial compensation programs already exist, management agencies or non-profit organizations often have large databases of livestock depredation cases that can be used to model carnivore risk to livestock. The process of building and applying risk maps can roughly follow the steps outlined below: My PhD involved developing spatial risk models for tigers and leopards on livestock in Kanha Tiger Reserve, central India. I saw first-hand how intuitively managers could understand risk maps and how maps could help inform management actions (e.g. fencing out livestock in high-risk areas).
Based on my experience of how easy and inexpensive risk models and hotspot maps are to build and use, I encourage researchers and managers to explore the many applications of these maps for their work. In particular, future efforts should focus on: · Building and validating robust models across more areas of the world where carnivore attacks on livestock cause major problems for local livelihoods and carnivore conservation. · Integrating risk models into long-term management and monitoring to explore whether they can effectively reduce human-carnivore conflict. · Developing new and innovative methods for displaying, sharing and applying results from risk models to reach new audiences (e.g. citizen science and smartphone apps). · Greater outreach with policymakers so that risk maps can inform large-scale decisions on conflict. · Investigating ecological and behavioral feedbacks that may occur after livestock grazing or patterns protection efforts change (would make a fantastic PhD project!). Spatial risk models and hotspot maps offer a simple, quantitative and insightful tool for the conservation toolkit. With greater and more widespread usage, risk models and maps will continue to improve the effectiveness of mitigation efforts for reducing livelihood losses and strengthening carnivore conservation worldwide. For more information, read the article: Miller JRB. 2015. Mapping attack hotspots to mitigate human-carnivore conflict: approaches and applications of spatial predation risk modeling. Biodiversity and Conservation (early online). PDF available here.
39 Comments
-- Colin Donihue PhD candidate It's hard to believe but I'm sitting on the island of Naxos straightening out logistics for my final field season in Greece! This is my fourth summer in the Cyclades and so far I've managed to avoid all of the logistical mistakes of previous years. I suppose the benefit of experience is the thrill of making brand new mistakes! Now that research equipment is all in one place, I'm pretty well settled in the 15th century castle I'm calling home for the next two months. (yeah, I really lucked out on that front!) This summer is going to be all about filling in the blanks. My field assistants and I had an epic field season last year which has put me pretty close to the mark for data collection. A winter's worth of data analysis though brought up some new questions about my little lizards, so that's the goal for this summer. Right now I'm working on untangling some inter-island differences in lizard bite force - an important lizard trait related to both diet and competition. I'm also finding some strong inter-population context-dependence on the big island of Naxos. I'll save the punchline for after the data is in one place, but I'm hoping this summer to be able to tie together a story about lizard hunting mode, morphology, performance, and behavior across the Greek landscape.
I'm also working hard to share stories and pictures from the field. I'll be updating my personal blog as often as possible. Find it here. Or find me on Twitter or Instagram under @colindonihue.  There's something new happening over on our Pubs Page! From here on out, whenever we publish a paper, we're going to include a short 50-word summary targeted towards the general public. Just click those "50 words" icons next to a paper and you'll be able to read the summary. Over the next few weeks we'll be going back through old publications to fill out the list, but we're excited about this challenge and opportunity to try to bring our research to new audiences. Readers: what do you think? Any ideas for further improvement? Other scientists: Will you try to do the same on your lab websites? We're looking forward to getting more summaries up on the site, and we'd love to hear what you think in the comments below. -- Jennie Miller, PhD '15 Originally published in the Yale F&ES Blog There are now fewer tigers in the wild than there are graduate students at Yale. Around 3,500 tigers, to be precise, dispersed through nine countries across Asia, and declining by the day. One of the greatest threats to tigers is retaliation from villagers after attacks on livestock. But to be fair, living with tigers in your backyard isn’t easy. Tiger attacks on livestock cause major income losses for pastoralists – up to 80% in some villages – threatening people’s livelihoods and personal safety. Yet this chain of conflict – cat kills cow, cow’s owner kills cat – is quickly emptying the jungles of the world’s most magnificent carnivore. In an effort to curb this human-tiger conflict, I focused my PhD on developing a tool to predict future attacks on livestock as a guide for grazing management and carnivore conservation. Specifically, I explored where and why tigers attack livestock, research that was recently published in Ecology and Evolution. I carried out my work in Kanha Tiger Reserve in Madhya Pradesh, central India. With its dense forests, Kanha serves as a stronghold for tiger conservation and is home to one of India’s largest tiger populations (around 70 individuals). Yet this booming source of tigers is a double-edged sword for local residents. The park’s ‘hard’ edge, formed by thick forests meeting sprawling agricultural fields, creates an abrupt interface between tigers and domestic animals where livestock are attacked daily. To assess tiger hunting I went straight for the kill – dead livestock – and investigated more than 400 animals killed by large carnivores around Kanha Tiger Reserve in central India. Using these kill sites, I built a spatial ‘risk model’ to identify the landscape features where tigers targeted livestock and map where future kills would occur. I found that tigers avoided human-dominated areas, like agricultural fields, and favored thick jungle where they could ambush grazing livestock. The risk of a tiger killing livestock increased near dense forests and away from roads, villages and open vegetation. As expected for a stalking carnivore, tigers killed livestock at sites with denser, patchier and more complex vegetation with low visibility. 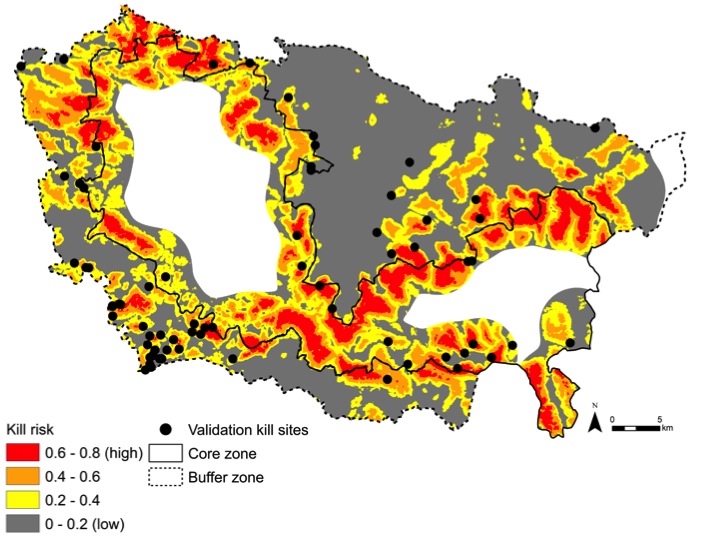 This risk map shows the relative probability of a tiger attacking livestock in Kanha Tiger Reserve, India. Risk models like these may assist managers and livestock owners in guiding livestock grazing away from high-risk habitats where tigers are most likely to attack, such as the dense forests around the park core zone boundary. Figure from Miller et al. 2015. Model predictions at a landscape scale showed high-risk livestock depredation hotspots inside the park core zone boundary, where livestock grazing is banned but still occasionally occurs. Hotspots were also located in several forest patches in the human-dominated buffer zone of the park.
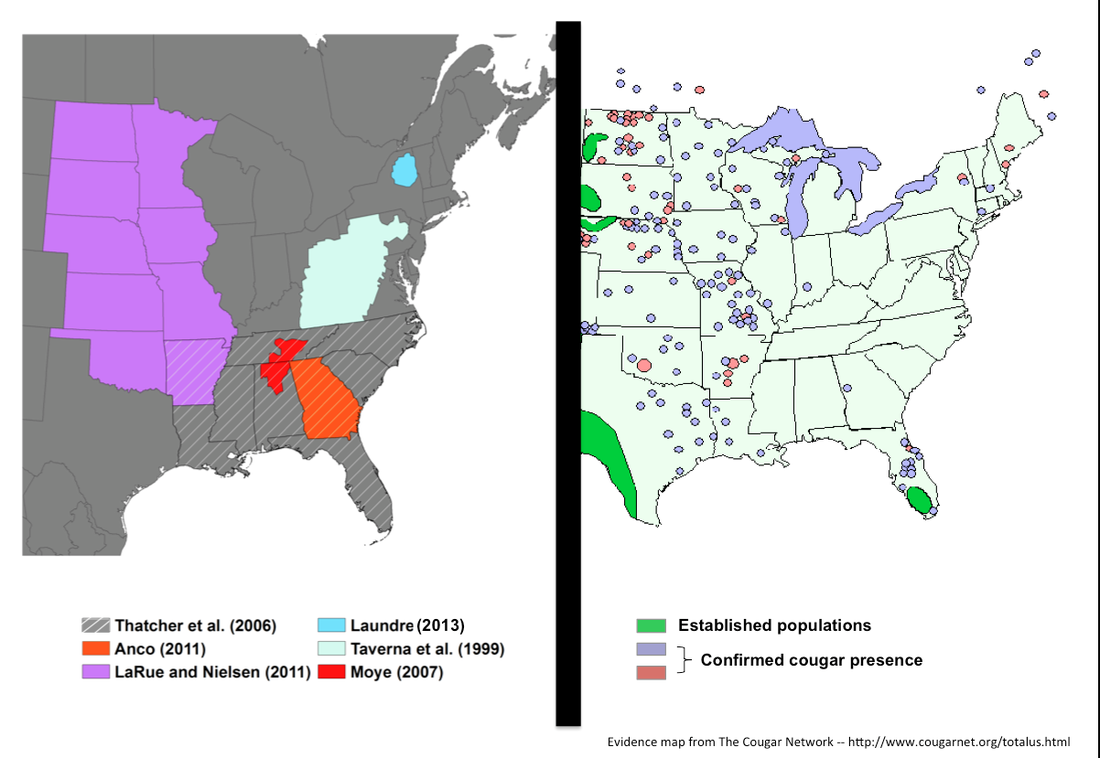
In working with park staff, I found that these risk hotspot maps could be an appealing visual tool for helping wildlife managers and livestock owners adapt grazing practices to minimize attacks. By grazing livestock in low-risk areas, such as open vegetation where tiers are less likely to attack, herders may be able to reduce livestock losses. Risk maps may also be useful for managers for targeting mitigating techniques, like fencing and awareness campaigns, to communities where tiger threats are highest. When combined with financial compensation to subsidize pastoralists incomes, these adaptive measures could offer simple, science-based practices for mitigating human-carnivore conflict. Additional research is still needed to explore the full utility and ramifications of using risk models for guiding management. Nonetheless, I feel hope that innovative, applied tools may offer new methods for enabling people and tigers to share landscapes. If so, then there might be a day in my lifetime when Yale graduate students are the rarer species. Research article: Miller, J.R.B., Y.V. Jhala, J. Jena and O. J. Schmitz. 2015. Landscape-scale accessibility of livestock to tigers: implications of spatial grain for modeling predation risk to mitigate human-carnivore conflict. Ecology and Evolution 5(6):1354–1367. -- Henry Glick, 2012 Master's Student After several hours combing through a giant stack of reports pulled from a file box at the Massachusetts Division of Fisheries and Wildlife’s Field Headquarters, it was clear that hardly a month goes by without someone reporting a cougar. No, I’m not talking about a cuddly kitten or fuzzy lap cat, but a wild cougar—a mountain lion who makes its living from the land, dragging down full-grown deer with jaws that can split vertebrae. Here in the Northeast, where large carnivores have been almost entirely absent for about a century, the thought of wild cougars roaming the patchwork landscape has raised a wide range of sentiments. For many, the idea sparks curiosity or a wash of romantic nostalgia for the wildness of yesteryear. For others, this idea elicits fear—fear that an afternoon walk through the park might turn them into an extra-large serving of Fancy Feast. The stack of cougar reports is maintained by Dr. Tom French, Assistant Director of Massachusetts’ Natural Heritage and Endangered Species Program and one of the region’s foremost students of cougar sightings. Compiled over many years by French and his colleagues, the stack contains records of not ten, twenty, or even fifty eyewitness accounts of cougars. No. This pile has nearly 2,000 reports scattered across the region, and they all tell the same story: cougars… cougars all over the place. This may be an exciting story, but it’s also a bizarre one considering that the in 2011 the U.S. Fish and Wildlife Service deemed the Eastern cougar extinct. Confusion about the presence of Puma concolor abounds. This is largely because those who have seen cougars—the believers—and those responsible for cougar management—the fish and wildlife agencies—aren’t seeing eye to eye. The believers are local residents like you or I, from all different walks of life. It has been through their observations that they have come to believe, relying on often fleeting glimpses of tawny fur to give them certainty. There’s just one problem: the regional fish and wildlife biologists, those tasked with managing the region’s natural resources (including such rare species as the cougar), have their doubts about what the believers have actually seen. The believers tell stories of terse conversations with incredulous biologists who raise their eyebrows at even the slightest mention of cougars. When authorities choose not to accept claims that four-legged eviscerators have begun to prowl the patchwork forests and suburban yards of New England, the believers become frustrated. The press feeds off this frustration, reporting believers’ claims alongside biologists’ disclaimers, while Internet message boards record unfiltered banter about the biologists’ managerial incompetence and organized deception. Fish and Wildlife agencies view their interactions with the believers from a decidedly different vantage. They assume that cougar sightings are erroneous unless proven otherwise – an approach rationalized by science and projected with an air of skepticism. Having chased down countless false reports, unless an actual animal turns up or laboratory-derived DNA points to cougars, biologists are reluctant to acknowledge the presence of the occasional animal that strays through the region, let alone a stable breeding population. The Northeast’s cougar controversy is confusing, not only because the evidence often falls within a grey zone of human interpretation, but also because the animals themselves are unpredictable. Take, for example, the first cougar carcass recovered from New England in the last 75 years, which came in 2011 after an unfortunate meeting of bumper and fur on the side of a parkway in Milford, Connecticut. As a town of 50,000 situated along the well-developed shoreline of the Long Island Sound, a mere 70 miles from the 8.2 million residents of New York City, Milford is hardly the wilderness one might have imagined. These incongruities of the cougar controversy were what first led me to Tom French’s office and ultimately to examine the Northeast’s capacity to support large carnivores. While much work has been done to examine evidence of cougars in the region—most notably a U.S. Fish and Wildlife review led by Endangered Species Biologist Mark McCollough (2011)—little has been done to evaluate cougar habitat. Researchers have used geo-spatial models to capture the defining characteristics Puma concolor habitat across the U.S. – California, South Dakota, Tennessee, Florida. However, the Northeast, which boasts the largest concentration of validated cougar evidence within the now extinct Eastern cougar’s historic range, had received little attention beyond Laundré’s (2013) efforts.  A cougar habitat distribution model in which “ideal” habitat characteristics have been derived from sites where cougars have been reported. Here, the Mahalanobis distance is used to quantify the degree of similarity between each location on the landscape and the pooled characteristics of reported locations. Dark green represents greatest similarity while dark red represents greatest dissimilarity. Habitat distribution models for generalist species like the cougar are of limited utility since there are few places these animals can’t live. However, where some information is better than no information, these models serve as valuable heuristic tools as management agencies continue to investigate dozens of eyewitness accounts each year. Through my work I found that the Northeast has, even under the most conservative of scenarios, enough habitat to support one or more small breeding populations of cougars. However, there are not, as far as we can tell, resident individuals in the region. Will there ever be? With the active expansion of cougar habitat into the Midwest, it’s likely well see more long distance dispersers in time. But while the region could support cougars from an ecological standpoint (i.e. plenty of space and plenty of food), the social carrying capacity may limit natural repopulation well into the future.
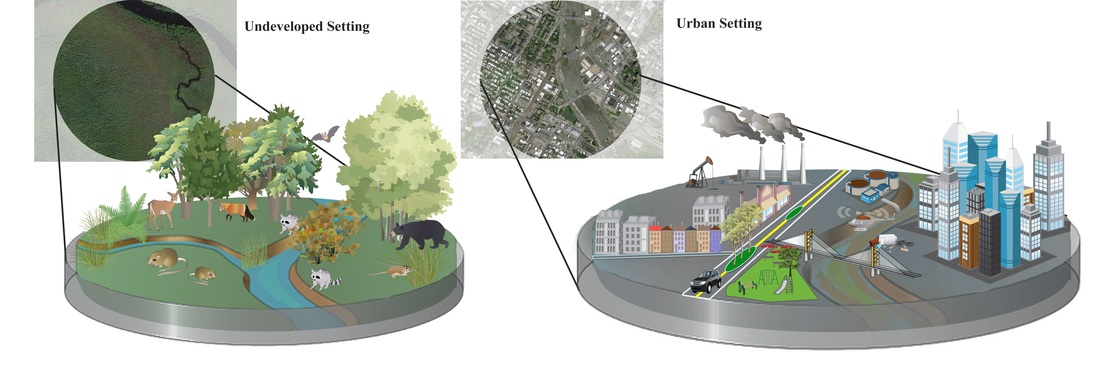
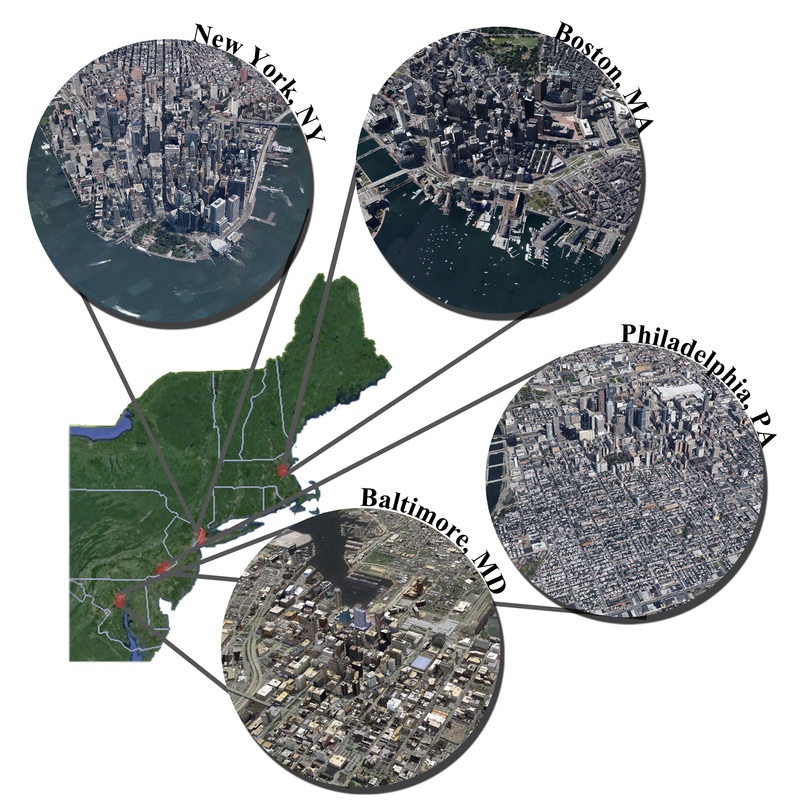
Want to learn more? Check out: Glick, H. B. 2014. Modeling cougar habitat in the Northeastern United States. Ecological Modelling, 285:78-89 [link URL: http://www.sciencedirect.com/science/article/pii/S0304380014000891] -- Alexandria Moore PhD Student It's hard to feel good when you first start a PhD program. Now, I don't know if that's true for everyone, but it was certainly true for me. The first year is filled with firsts: First time moving away from home, first time developing a research project, first time being part of a community, first time recognizing that everyone has moments where they don't feel good enough. The hardest first for me was realizing that I don't always know how to conquer those not-good-enough [to develop my own research, to make friends, to be here] feelings. The PhD struggle is less about the work and more about how you are able to provide yourself with enough support to do it and feel good about it. During my first year, I found support in my cohort, in my lab mates, and in my dedication to developing a research project that I cared enough about to spend years on. Of these, my research has been the mainstay of my support structure. I entered Yale with a passion for conservation, but little sense of direction. As I navigated my first semester as a doctoral student, my research project began to take shape and, because this is a science blog, I'm going to share the progress I made in Year One. I work in wetlands. They're dirty, they smell, and they're often covered in crabs, but given the right perspective, they're beautiful. Wetlands are also an invaluable ecosystem type in decline worldwide. Since the 1980s, restoration projects have been implemented to return degraded landscapes back to their natural states. However, restored wetlands tend to exhibit reduced structure and function compared to natural wetlands. This is where my research comes in. I am interested in taking an ecological perspective to evaluate why restored wetlands don't function as well as natural ecosystems. In particular, I ask: Are species diversity and community structure important for ecosystem functioning? Do the presence of predators, herbivores, and plant species impact not only the biotic community, but larger ecosystem-level processes? To begin looking at this question, I selected three salt marshes (one natural, one restored, and one degraded) along the New England coast and collected baseline data on ecosystem functions and biodiversity. Preliminary results show that, as expected, the restored marsh has reduced functioning compared to the natural site. But the question of how the ecological community affects this is, as of yet, unaddressed. I've only completed my first summer, so I don't have an answer, but in preparing for summer number two and years 3, 4, and 5, I'm optimistic that I'll get close to one. The nature of a PhD, at least the nature of mine, is that it's hard to really feel sure of anything. But between the days when I don't feel good enough and the days when I do, I'm optimistic. To bring things back to where they started: It's hard to feel good when you first start a PhD program, unless you have support from within yourself and from outside of yourself. For me, that support hasn't been constant and there were plenty of mad/sad/what-am-I-doing-here days, but the happy/hopeful/this-is-what-I-want-to-do days made the other kind tolerable. Entering Year Two, I still have plenty of both types of days, sometimes more of one than the other, but I accept them as they come. The first year is a pivotal one for recognizing that although getting a PhD is a struggle, you're one in a community of others struggling alongside you - sometimes happy, sometimes sad, but often optimistic. -- Colin Donihue PhD Candidate Humans are rapidly changing landscapes, but the increasing realization that species can rapidly adapt to their dynamic surroundings has led to the intriguing possibility that ecological systems may be more resilient to human land use than previously appreciated. The processes governing the adaptability of species in human contexts are still relatively unknown as the discipline's historical modus operandi (think Darwin) has been to study evolution in more natural or laboratory contexts. However, humans are one of the most potent drivers of adaptation (Hendry and Kinnison 1999), so it makes sense to study those adaptive dynamics where humans are most prevalent and ecological stressors most varied, abundant, and novel: cities. A fellow student, Max Lambert, and I just wrote a perspective article for AMBIO* making this case. Let me elaborate a bit more on our argument. The field of urban ecology has flourished in the last couple of decades. Within that time it has made several exciting discoveries leading to fundamental paradigm shifts within the discipline. Where scientists first focused on ecology in cities (e.g., distribution of raccoons relative to trash) they have now shifted to studying the ecology of cities (e.g., heat island effect), explicitly acknowledging the inextricable role of humans in the dynamics of the system. As numerous studies have convincingly demonstrated (see the Phoenix and Baltimore LTERs), cities are functional ecosystems with unique and characteristic ecologies. We argue then that it follows that the species composing those communities would be evolving to their novel and dynamic contexts. Tracking evolution and testing for adaptive trait changes in city species requires a multi-step research program. Initially, identifying traits that vary with environmental context will be facilitated by the gradients that characterize many of these potential adaptive drivers in urban settings (thermal, food, noise, habitat structure, chemical; Fig. 1). However, context-dependent trait variation does not necessarily imply adaptation, so subsequent common garden experiments or reciprocal transplant experiments can give insight to the genetic basis and fitness implications of the observed trait variability. These experiments are often logistically complicated in city centers, but already great examples exist of creative experiments that begin exploring these questions. Why is this important? Studying evolution in urban contexts enables new insight difficult or impossible to gain in more natural settings on the adaptive potential of species caught in humanity's headlights. We propose that species in cities may be evolving faster than previously appreciated, in time with the fastest rates of urbanization in human history (Seto et al. 2010). Furthermore, because cities often encompass predictable and comparable gradients in many of these potential adaptive drivers (Fig. 1), comparing the evolutionary trajectories of urban species in multiple cities (Fig. 2 and manuscript text) will give great insight to the question of repeated and therefore predictable evolution, a fundamental point of debate since Darwin. Urban planners are calling on ecologists to help design urban environments to better achieve conservation goals (Felson et al. 2013). Understanding the cause, rate, and predictability of species adaptation to humans will better inform urban design, species conservation, and our basic understanding of one of biology's few constants, evolution. Want to read more about applied work going on in Urban Ecology and Landscape Architecture? The Yale Hixon Center is doing some really exciting work.
A great deal of work by other researchers is generally referenced in this post. Please see published paper for full citations and attribution. * If you can't access the paper please email me for an uncopyrighted draft of the manuscript. Works cited: - Felson, A.J, E.E. Oldfield, and M.A. Bradford. 2013. Involving ecologists in shaping large-scale green infrastructure projects. BioScience 63: 882-890. - Hendry, A.P., and M.T. Kinnison. 1999. The pace of modern life: Measuring rates of contemporary microevolution. Evolution 53: 1637-1653. - Seto, K.C., R. Sanchez-Rodriguez, and M. Fragkias. 2010. The new geography of contemporary urbanization and the environment. Annual Review of Environmental Resources 35: 167-194. -- Jennie Miller PhD Candidate  Hunting_Cripple Creek Colorado In suburban neighborhoods across the globe, it is no longer surprising to see deer grazing outside downtown offices in the mid-day sun, brazenly snacking along sidewalks or grazing on gardens as pedestrians pass within petting distance. These animals, once cherished for their shy and flighty nature, now boldly roam among humans and their property, spreading disease and environmental devastation in their wake. Managers have had little success in their attempts to control overabundant ungulates with common methods such as culling and contraceptives, which are also costly and controversial. However, a recent review published in the Journal of Applied Ecology presents a new wildlife management tool that may offer a less bloody and more effective alternative: fear. Over the past decade, ecologists have found mounting evidence of an idea that horror films have exploited over the last century – that the threat of death is scarier than death itself. Just as countless starlets have cowered from mere threats of torture from dark-hooded movie murders, prey animals make great sacrifices to avoid the simple threat of attack from wild carnivores. Just the presence of predators – be they tarantulas, terns or tigers – can elevate stress levels in their prey that hamper feeding, reproduction and ultimately survival. In essence, predators terrify their prey long before they pounce. The non-lethal ‘fear effects’ from carnivores can constrain prey populations as strongly as – if not stronger than – actual kills made by carnivores. Yet most management programs aimed at reducing overabundant deer populations have ignored fear effects, instead using methods such as hunting and birth control that rely on mortality alone to control ungulates. In their March 2013 article, Dr. Joris Cromsig and colleagues highlight the importance of incorporating fear effects into ungulate management. Humans should, basically, scare ungulates to death. Cromsig et al. suggest adapting current practices of hunting to generate greater fear in ungulates. By extending the hunting season and area, hunters could evoke greater and more constant levels of fear in ungulates, creating stress that reduces herd sizes and shifts grazing patterns. But can humans adequately fill the role of large carnivores and limit ungulate populations through fear alone? As a doctoral student who spends hours contemplating how fear affects prey, I found Cromsig et al.’s suggestion intriguing yet in need of additional discussion. In applying ideas about the ‘ecology of fear’ to practical management, Cromsig and colleagues contribute to an emerging conversation on predation risk as a management tool (for examples see Berger 2007 and Treves et al. 2011). Non-lethal fear effects do appear to have great – and largely unexplored – utility for wildlife management issues such as ungulate overgrazing, livestock depredation and carnivore conservation. And by infusing fear effects into current practices of hunting implemented in countries worldwide, Cromsig et al. present a technique that could generate drastic changes with minimal cost and effort.  Deer_Wodu Media While the theoretical framework is strong, several limitations offer challenges in implementing the idea of ‘hunting for fear’ as a management tool. For one, ecologists have yet to fully understand how the fear of carnivores varies spatially and temporally, as well as with predator hunting mode, so simulating fear effects in nature would be difficult. Furthermore, because game management involves humans as much as animals, hunters would need to adapt their fear effects uniquely by location to meet both cultural expectations and ecosystem composition. Cromsig and colleagues recognize these challenges and discuss them in their article. However, several additional issues would be valuable to consider when deciding whether and how to implement ‘hunting for fear’ for ungulate control. First, increasing human activity in natural spaces – even as a management tool – must be scrupulously considered because these efforts could easily result in unintended side effects on the ecosystem. Recreating carnivore fear effects with humans would be most applicable in areas where carnivore populations have been extirpated or reduced to densities where predators no longer generate significant fear effects on prey populations. If predators are still present in the environment (and even if they are not), the presence of humans could strongly disturb the ecosystem, creating more problems than solutions. Most carnivores, as well as numerous other species, are highly sensitive to human presence and spatially and/or temporally avoid human activity, altering their behaviors and roles within the food web. These types of unintended side effects on the ecosystem must be examined carefully prior to implementation to determine whether ‘hunting for fear’ could do more harm than good. Second, even with a low predator density, encouraging humans to spend more time in wild spaces might inflate human-wildlife conflict in parts of the world. For instance, bringing humans into jungles with low densities of tigers and leopards in an attempt to control their populous prey species (such as wild pigs and nilgai antelope, which are considered some of the worst crop-raiding pests in southeast Asia) could displace big cats from their last remaining habitats, cause them to hunt livestock and/or encourage attacks on people. At the least, increasing the chance of confrontation between humans and large carnivores could likely further amplify negative attitudes towards carnivores that already create challenges for conservation. Thus, ‘hunting for fear’ should likely not be attempted in locations where dangerous large carnivores still exist, even in low densities. Considering these concerns, it appears that ‘hunting for fear’ might generate the greatest ecological benefits with the fewest negative repercussions on humans and wild species alike in areas where carnivores have been extirpated and ungulate control is desperately needed to restore ecosystem health. Prime examples include the urban areas of many developed countries in Europe and North America, such as the United States where Cromsig et al. point to over-browsing by white-tailed deer (Odocoileus virginianus) as the cause of the extinction of species such as White trillium (Trillium grandiflorum ). In these areas where large carnivores are locally extinct, people could functionally replace the fear effects on prey that carnivores once created when food webs were intact. Indeed, this possibility speaks to the powerful role that humans can play in the ecosystem and illustrates the importance of a new perspective that incorporates humans into food webs, such as human-nature coupling (Liu et al. 2007). The incorporation of fear into hunting as a management tool requires careful forethought and small-scale experimentation to avoid devastating side effects, such as those that have accompanied the introduction of invasive species to control agricultural pests. I encourage particular forethought prior to the application of ‘hunting for fear’ in countries with poor forest management infrastructure, where hunting may be especially difficult to regulate. Overall, incorporating fear effects into hunting may offer an innovative technique for controlling ungulate populations. ‘Hunting for fear’ is a prime example of a practical tool that applies principles from species interactions to solve an ecological problem. These types of tools are rare yet essential if science-informed management is to rigorously address the complexities of species and ecosystem conservation. -- Adam Rosenblatt Post-doc  Photo Credit: Axel Schulz Photo Credit: Axel Schulz Catching caimans is not that complicated. First, you get in a boat at night, head out on the river, and use high-powered spotlights to find the animals. The light from the spotlights bounces off the tapetum lucidum (a layer of tissue within the retina that reflects light back into the eye, increasing the clarity of night vision), making the caiman’s eyes shine bright red from hundreds of meters away. Second, you drive the boat slowly towards the caiman, making as little noise as possible (crocodilians have good hearing). Third, a person at the bow of the boat uses a long pole to slip a metal snare around the neck of the caiman, and then tightens the snare by pulling on the rope it’s attached to or by yanking the pole upwards swiftly. Fourth, you hold on for dear life as the caiman thrashes around in the water, tiring itself out. Crocodilians tire relatively quickly (5 minutes or so) during bursts of intense physical activity because lactic acid, a natural by-product of cellular metabolism, builds up in their muscles and shuts them down. Human muscles also suffer from lactic acid build-up during exercise, but our muscles are able to get rid of it relatively quickly while crocodilians are stuck with it for longer periods of time because of their cold-blooded physiology. Fifth, you use a second snare to close the mouth and secure it, then you tape the mouth shut. Now you’re ready to safely handle the animal. During my time in Guyana my local counterparts and I caught 30 adult caiman this way, and to find out what they were eating we pumped their stomachs. Instead of explaining the whole stomach pumping process, you can watch this short video I made: https://www.youtube.com/watch?v=JCmogf-NQ2Y We ended up finding lots of interesting stuff in their stomachs, like snails, fish, seeds, and small mammals. Fish were by far the most frequently consumed type of prey, which means in the region I was in fishermen and caimans directly compete for food, at least during the dry season. The long-term challenge is to figure out how, despite this overlap, to maintain the quality of life for humans who depend on fish for food while also protecting the caiman population and the important roles caiman play in local food web and ecosystem dynamics. My goal is to go back to Guyana in the coming years to continue and expand upon the caiman research I began in February.
-- Adam Rosenblatt Post-doc Some research sites are really hard to get to. Back in 2007 I worked for 6 months in Shark Bay, Australia as a volunteer on Mike Heithaus’ long-running research project, and getting there was a schlep. I had to fly from Philly (where I lived at the time) to LA, then to Auckland, New Zealand, then to Perth, Australia, then take a bus 10 hours up the Australian west coast to a gas station in the middle of nowhere called “The Overlander,” then take another bus for an hour to the research site, Monkey Mia. By the end your bones ache and your eyes are weary with uncomfortable fits of sleep, but the beauty and possibility of such a remote place overtakes you and quickly you forget about the hardships of the journey. I recently did some research in another remote part of the world: Guyana. It’s a country few have heard of, tucked away on the northern coast of South America, dwarfed by its giant neighbor Brazil. Yet Guyana boasts vast tracts of untouched forests and savannahs, and some of the highest biodiversity on the planet. Guyana’s wilderness is impressive and dense, and it has remained largely undeveloped precisely because not many people know where it is or are aware of its natural splendor. I headed down there, to Guyana’s interior savannahs, to do some research on the largest crocodilian in the western hemisphere: the black caiman. To get there, I flew from New Haven to Miami where I picked up some supplies and equipment, then on to Port of Spain, Trinidad, then on to Georgetown, Guyana’s capital city. After a few days spent in Town (as the locals call it) to get my research permits, I hopped on a tiny little plane that flew me to a dirt airstrip near the Brazilian border where I was picked up by my local research collaborators in a red dust-covered pick-up truck. We drove 45 minutes over rain-scoured dirt roads and through a 3 mile long forest to the village of Yupukari, which sits on a hill above the Rupununi River and Awarikuru Lake. The Kanuku Mountains are located just to the south, and you can easily see them from Yupukari. The view is particularly beautiful in the evening when the mountains take on a purplish glow as the sun sets. The village is made up of about 1000 Macushi Amerindians, one of the indigenous groups in the region. It’s a fishing village in the dry season and a hunting village in the wet season, and I was there to figure out what black caimans eat and what ecological roles they play in the area. My base of operations was the lovely Caiman House eco-lodge, a popular place for tourists to hang their hats as they explore the amazing birds, fish, reptiles, and mammals of the region. When I arrived I unpacked my things in my breezy room, checked the integrity of the mosquito net hanging over my bed, and headed to the kitchen for a meal of fresh-caught fish, rice, and the spiciest chili peppers I’ve ever encountered. I was in paradise, or at least one version of it, and I was excited to see what surprises the surrounding waters held in store for me.
Next time: Caimans, and lots of them. |
Archives
November 2019
Categories
All
|















 RSS Feed
RSS Feed
